REVIEW ARTICLE
TECHNOLOGIES FOR INTRAVENOUS DRUG ADMINISTRATION: A SCOPE REVIEW
TECNOLOGIAS PARA A ADMINISTRAÇÃO DE MEDICAMENTOS POR VIA INTRAVENOSA: UMA REVISÃO DE ESCOPO
TECNOLOGÍAS PARA LA ADMINISTRACIÓN DE MEDICAMENTOS INTRAVENOSOS: UNA REVISIÓN DEL ALCANCE
https://doi.org/10.31011/reaid-2026-v.100-n.2-art.2587
José Mateus Ismael Lima1
Fernanda Eli Dantas Gondim2
Bernadete de Lourdes André Gouveia3
Maria Cristina Lins Oliveira Frazão4
Luana Carla Santana Ribeiro5
1 Universidade Federal de Campina Grande. Cuité, Paraíba, Brazil. https://orcid.org/0009-0009-7650-8568
2 Universidade Federal de Campina Grande. Cuité, Paraíba, Brazil. https://orcid.org/0009-0003-1065-1819
3 Universidade Federal de Campina Grande. Cuité, Paraíba, Brazil. https://orcid.org/0000-0003-3485-3100
4 Universidade Federal de Campina Grande. Cuité, Paraíba, Brazil. https://orcid.org/0000-0001-8133-6048
5 Universidade Federal de Campina Grande. Cuité, Paraíba, Brazil. https://orcid.org/0000-0002-8069-5492
Corresponding Author
José Mateus Ismael Lima
Rua José Janduir Carneiro, 124, Bairro Pref. Jaime Pereira, 58175000, Cuité-PB, Brazil. Phone: +55(83) 991687822. E-mail: ismaeljosemateus@gmail.com
Submission: 31-05-2025
Approval: 04-12-2025
ABSTRACT
Introduction: Intravenous medication administration is an essential and complex practice, requiring attention, qualification and use of technologies to promote patient safety. Objective: This study aimed to map the scientific evidence and technologies on intravenous medication administration, performed by nursing professionals, within the scope of health services. Methods: This is a scoping review carried out in accordance with the recommendations of the Joanna Briggs Institute, conducted with studies from the MEDLINE, LILACS, BDENF, Web of Science, Scopus and CINAHL databases, in addition to theses and dissertations repositories. Studies in the time frame from 2017 to 2024, which address educational technologies, devices and techniques related to intravenous administration, were included. Results: Three main categories were highlighted that guided the data analysis, related to the techniques/devices involved in intravenous administration; common errors in intravenous administration and prevention measures; and educational technologies used in the training of professionals focused on intravenous administration, identifying innovations in medical devices, educational tools and operational protocols that aim to reduce errors and improve efficiency in the intravenous administration process. Furthermore, the analyses emphasized the importance of continuous training and the use of evidence-based methodologies to prevent complications and improve the quality of care. Conclusion: it is understood that the adoption of technologies and continuous training is essential to improve clinical practice and reduce risks associated with intravenous administration. This study reinforces the need to integrate scientific evidence into daily care, ensuring quality and safety in health care.
Keywords: Educational Technologies; Intravenous Administration; Nursing.
RESUMO
Introdução: A administração de medicamentos intravenosos é uma prática essencial e complexa, exigindo atenção, qualificação e uso de tecnologias para promover segurança ao paciente. Objetivo: Este estudo objetivou mapear as evidências científicas e tecnologias sobre a administração de medicamentos por via intravenosa, realizada por profissionais de enfermagem, no âmbito dos serviços de saúde. Métodos: Trata-se de uma revisão de escopo realizada conforme as recomendações do Joanna Briggs Institute, conduzida com estudos das bases de dados MEDLINE, LILACS, BDENF, Web of Science, Scopus e CINAHL, além de repositórios de teses e dissertações. Foram incluídos estudos no recorte temporal de 2017 a 2024, que abordam tecnologias educacionais, dispositivos e técnicas relacionadas à administração intravenosa. Resultados: Foram destacadas três categorias principais que orientaram a análise dos dados, relacionadas às técnicas/dispositivos envolvidos na administração intravenosa; erros comuns na administração intravenosa e medidas de prevenção; e tecnologias educacionais utilizadas na capacitação dos profissionais voltadas à administração intravenosa, sendo identificadas inovações em dispositivos médicos, ferramentas educacionais e protocolos operacionais que apontam para reduzir erros e melhorar a eficiência no processo de administração intravenosa. Ademais, as análises enfatizaram a importância da capacitação contínua e do uso de metodologias baseadas em evidências para prevenir complicações e aprimorar a qualidade do cuidado. Conclusão: compreende-se que a adoção de tecnologias e treinamento contínuo é essencial para melhorar a prática clínica e reduzir riscos associados à administração intravenosa. Este estudo reforça a necessidade de integrar evidências científicas ao cotidiano assistencial, garantindo qualidade e segurança no cuidado em saúde.
Palavras-chave: Tecnologias Educacionais; Administração Intravenosa; Enfermagem.
RESUMEN
Introducción: La administración de medicamentos intravenosos es una práctica esencial y compleja, que requiere atención, calificación y uso de tecnologías para promover la seguridad del paciente. Objetivo: Este estudio tuvo como objetivo mapear evidencias científicas y tecnologías sobre la administración de medicamentos intravenosos, realizada por profesionales de enfermería, en el ámbito de los servicios de salud. Métodos: Se trata de una revisión de alcance realizada de acuerdo con las recomendaciones del Instituto Joanna Briggs, realizada con estudios de las bases de datos MEDLINE, LILACS, BDENF, Web of Science, Scopus y CINAHL, además de repositorios de tesis y disertaciones. Se incluyeron estudios en el periodo de tiempo de 2017 a 2024, que abordan tecnologías, dispositivos y técnicas educativas relacionadas con la administración intravenosa. Resultados: Se destacaron tres categorías principales que orientaron el análisis de los datos, relacionadas con las técnicas/dispositivos involucrados en la administración intravenosa; Errores comunes en la administración intravenosa y medidas de prevención; y tecnologías educativas utilizadas en la formación de profesionales enfocados en la administración intravenosa, identificando innovaciones en dispositivos médicos, herramientas educativas y protocolos operativos que busquen reducir errores y mejorar la eficiencia en el proceso de administración intravenosa. Además, los análisis enfatizaron la importancia de la capacitación continua y el uso de metodologías basadas en evidencia para prevenir complicaciones y mejorar la calidad de la atención. Conclusión: se entiende que la adopción de tecnologías y la capacitación continua es fundamental para mejorar la práctica clínica y reducir los riesgos asociados a la administración intravenosa. Este estudio refuerza la necesidad de integrar la evidencia científica en la atención diaria, garantizando la calidad y seguridad en la atención sanitaria.
Palabras clave: Tecnologías Educativas; Administración Intravenosa; Enfermería.
INTRODUCTION
Medication administration is one of the most frequently performed activities during clinical practice, being a complex process that requires the attention and qualification of professionals involved in patient care. Among the routes of medication administration, the intravenous route, frequently used for drug infusion, constitutes a complex practice that involves risks for patients and professionals, commonly used to administer medications systemically and in a short period of time, requiring scientific knowledge and technical skill on the part of the professionals involved, to prevent iatrogenic effects associated with this process that compromise patient safety (1).
Since the initiative of the World Health Organization (WHO) in 2004, with the creation of the World Alliance for Patient Safety, strategies have been developed to improve patient safety (2). One of these strategies was the publication of the Global Patient Safety Challenge, with the aim of mapping risk areas and developing tools to prevent harm to patient safety. From this perspective, in 2017, the WHO published the third global challenge, called “Medications without data”, with the objective of reducing critical harm associated with medication administration errors by 50% in the coming years (3). Thus, this challenge highlights the extreme need to improve the quality and training of professionals during the medication administration process, aiming to promote patient safety.
Regarding medication administration, the nursing team plays a relevant role in ensuring the safety and quality of care. In light of this, in accordance with Resolution No. 564/2017 of the Federal Nursing Council, it is the responsibility of these professionals to guarantee care based on technical and scientific knowledge, so that, in medication administration, it is essential that they understand the therapeutic indication, the action of the drug, the route of administration and the possible associated risks, so that they are able to identify inconsistencies in the medical prescription, refraining from administering the medication until it is clarified with the prescriber, aiming to reduce the occurrence of harm and promote patient safety (4). Considering this context of practice, it is fundamental that nursing professionals seek continuous training, updating themselves, in order to mitigate possible harm to patient safety.
From this perspective, in the process of administering medications intravenously, the attention of professionals is extremely necessary to avoid complications during medication infusion. Given this scenario, an analysis of the main errors committed in the infusion of intravenous medications revealed that the main cause of errors is related to the technique adopted, both in the preparation (60%) and administration (75%) of medications via this route. Regarding preparation, it is evident that the main technical failures are associated with understanding the prescription, disinfection of the work surface and ampoules, hand hygiene, identification of prepared medications, use of PPE, and contamination of materials. In administration, technical errors involve understanding the prescription, patient identification and orientation, hand hygiene, checking the medication label, use of PPE, disinfection of the access device entry point, and contamination of materials (5).
From this perspective, currently, a tool to promote the minimization of such errors consists of the use of technologies that assist not only in the preparation and administration of medications and patient monitoring, but also in the training of the professionals involved. Along these lines, there are several technologies that ensure safer intravenous infusion practices, notably intravenous catheters with safety devices, vein visualization devices, intelligent infusion pumps, and innovative monitoring software (6). Regarding professional training, there are several educational technological innovations used in training, notably virtual reality simulators, realistic audiovisual productions, virtual learning platforms, games, websites, guides, illustrated manuals, among other technologies (7-10).
Given the immense diversity of technologies involved in healthcare practice and the development of global strategies to promote greater safety in medication administration, this study is justified by the need to gather and analyze the main scientific evidence regarding the use of technologies for intravenous medication administration, to provide an overview of the advances and challenges in promoting patient safety and minimizing errors associated with clinical practice, after the implementation of strategies defined by the UN.
Furthermore, a preliminary search conducted in the MEDLINE, Cochrane Database of Systematic Reviews, and JBI Evidence Synthesis databases did not identify current or ongoing systematic or scoping reviews addressing this topic, highlighting the knowledge gap and reinforcing the relevance of conducting a scoping review to map the scientific evidence in this field of knowledge.
Therefore, this study aims to map the scientific evidence and technologies regarding intravenous medication administration performed by nursing professionals within healthcare services.
METHODS
This is a scoping review, prepared according to the Joanna Briggs Institute (JBI) scoping review methodological guidelines. This type of research allows for a thorough mapping of the literature, aiming to provide a broader overview of the subject, and is conducted through five stages: determining the guiding research question; identifying relevant scientific evidence; selecting studies; evaluating the data obtained; summarizing and presenting the findings (11). Furthermore, the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA-ScR) checklist (12) was followed to guide the writing of the review.
The selection of this research method was considered relevant because it allows for an extensive literature search to identify scientific evidence and technologies related to the process of administering medications intravenously, within the scope of nursing professionals' work in health services.
To formulate the guiding research question, objective, and descriptors, the acronym PCC was used, identifying that P (Population) refers to nursing professionals (nurses, technicians, and nursing assistants); C (Concept) refers to technologies and scientific evidence regarding the administration of intravenous medications to young, adult, and elderly patients; and C (Context) refers to health services (Family Health Units, Emergency Care Units, hospitals, pre-hospital care, among others). Thus, the following research question was formed: “What technologies and scientific evidence are available regarding the administration of intravenous medications by nursing professionals in health services to young, adult, and elderly patients?”
This scoping review included studies that addressed technologies and scientific evidence regarding the administration of intravenous medications to young, adult, and elderly patients by nursing professionals within health services. In this sense, studies were selected that addressed: types of intravenous medications and solutions; Validated educational technologies (guides, manuals, videos, etc.) aimed at nursing professionals; techniques/procedures for intravenous medication administration; and devices/materials for intravenous medication administration, available in indexed journals, textbooks, dissertations and theses in established databases, care protocols/guidelines, with a time frame of the last 7 years, from 2017 to 2024, without language limitations.
The study's time frame is justified by the implementation, in 2017, of the third Global Patient Safety Challenge, launched by the World Health Organization (WHO) under the title "Medication Without Harm," aiming to reduce, within five years, 50% of serious and preventable harm related to the use and administration of medications. To achieve this goal, the challenge proposes the development and implementation of specific guidelines, documents, technologies, and tools to promote the safe use of medications. Thus, the temporal delimitation of this study is based on the mapping of technologies and products developed in response to this global WHO initiative.
For inclusion, observational studies with quantitative and qualitative approaches were considered, such as cross-sectional, cohort, and case-control studies; experimental studies, including planned and non-randomized clinical trials; quasi-experimental studies; and systematic reviews, meta-analyses, reviews with clinical protocols, or clinical guidelines. Regarding exclusion criteria, studies published in conference proceedings, letters to the editor or editorials, opinion articles, and literature reviews that did not follow systematic, integrative, or scoping formats were disregarded.
Regarding the search strategy, a two-step approach was used to locate both studies published in indexed journals and studies in the grey literature, following JBI recommendations (11). First, an initial search for descriptors was conducted using the Health Sciences Descriptors (DeCS), obtaining the terms organized based on the PCC acronym: Population (Nursing); Concept (Educational Technology; Biomedical Technology; Administration, Intravenous; Infusions, Intravenous; Catheterization, Peripheral); and Context (Health Services). Then, the descriptors were cross-referenced using the Boolean operators AND and OR, formulating three search strategies, described in Table 1.
Table 1 - Search strategies defined for the scoping review. Cuité, Paraíba, Brazil, 2024.
|
Search |
Strategy used |
Records found |
|
1 |
Nursing AND (Administration, Intravenous OR Infusions, Intravenous) AND (Educational Technology OR Biomedical Technology). |
129 |
|
2 |
Nursing AND (Administration, Intravenous OR Infusions, Intravenous) |
15195 |
|
3 |
Nursing AND Catheterization, Peripheral. |
1954 |
Source: Research data (2024).
From this perspective, the following databases were considered for obtaining published studies: Medical Literature Analysis and Retrieval – MEDLINE (via PubMed); Latin American and Caribbean Literature in Health Sciences – LILACS and Nursing Database – BDENF (both via Virtual Health Library – VHL); Web of Science, Scopus and CINAHL (via CAPES Journals Portal). Furthermore, a search for theses and dissertations was conducted in the repositories of the USP Digital Library of Theses and Dissertations and the Catalog of Theses and Dissertations of the journal of the Coordination for the Improvement of Higher Education Personnel (CAPES).
After searching the databases and repositories, the studies were included in a free web-based review software, the Rayyan Qatar Computing Research Institute (Rayyan QCRI) (13). In this program, duplicates were identified and removed. Following this process, two independent reviewers performed blind screening using Rayyan software, analyzing titles and abstracts to verify their suitability to the review's inclusion criteria.
Subsequently, the pre-selected full texts were analyzed, based on the inclusion and exclusion criteria, again by two independent reviewers. The results of the analyses were compared, with any discrepancies between the reviewers resolved by a third additional reviewer. This process took place between August and December 2024, and the results of the search and inclusion process are presented in the PRISMA flowchart, as shown in Figure 1 (12).
Figure 1 - Flowchart of the search and selection stages of studies for the development of the scoping review. Cuité, Paraíba, Brazil, 2024.
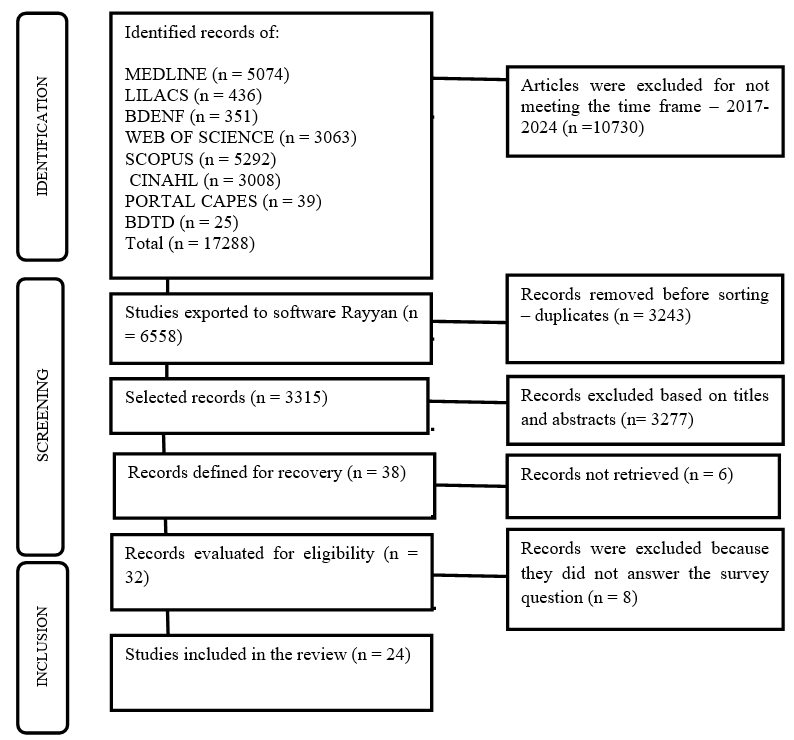
Source: Research data (2024).
To extract data from the articles included in the scoping review, the tool proposed by JBI, adapted to the research objective, was used (11). In this sense, the following information was extracted from the studies: title, authors, year of publication, country of origin, database, journal, population, sample size, age range of participants, study objective, type of study, sample and data analysis, as well as the main findings, highlighting the type of educational technology, techniques/procedures and devices/materials related to intravenous administration reported in the study, the main results and conclusions.
Finally, the results underwent a meticulous interpretation, followed by synthesis and discussion based on the selected scientific literature. This process was conducted using Microsoft Office Word software.
RESULTS
After analyzing the content of the studies selected for this scoping review on technologies for intravenous drug administration, a synthesis of the data contained therein was developed, regarding title, authors, database, journal, year, country of publication, and language, which is presented in Table 1.
Table 1 – Characterization of the articles selected for scoping review. Cuité, Paraíba, Brazil, 2024.
|
Article number |
Title |
Authors |
Database |
Períod |
Year |
Country |
Language |
|
A1 |
Processo de inserção, manutenção e retirada de cateter intravenoso periférico: análise preventiva de riscos(14) |
Furlan M da S, Saba A, Berger S, Lima AFC. |
SCOPUS |
Cogitare Enfermagem |
2023 |
Brasil |
Português |
|
A2 |
Protótipo de tecnologia de cuidado para prática do flushing na manutenção de cateteres intravenosos(15) |
Ribeiro GSR, Campos JF, Parreira PMSD, Silva RC. |
LILACS |
Texto & Contexto Enfermagem |
2023 |
Brasil |
Português |
|
A3 |
Administração segura de medicamentos via endovenosa: uma proposta para o hospital universitário(16) |
Alves SZSP |
LILACS |
PPGSTEH – UNIRIO |
2017 |
Brasil |
Português |
|
A4 |
Pain and Satisfaction Perceptions of Ultrasound-Guided Versus Conventional Peripheral Intravenous Catheterization: A Randomized Controlled Trial(17)
|
Salleras-Duran L, Fuentes-Pumarola C, Fontova-Almató A, Roqueta-Vall-Llosera M, Câmara-Liebana D, Ballester-Ferrando D. |
PubMed |
Pain Management Nursing |
2024 |
Estados Unidos |
Inglês |
|
A5 |
Validação de instrumento para intervenção de enfermagem ao paciente em terapia vasoativa(18) |
Paim AE, Nascimento ERP, Bertoncello KCG, Sifroni KG, Salum NC, Nascimento KC. |
PubMed |
Revista Brasileira de Enfermagem |
2017 |
Brasil |
Português |
|
A6 |
A Simulation-Based Blended Curriculum for Short Peripheral Intravenous Catheter Insertion: An Industry-Practice Collaboration(19) |
Glover KR, Stahl BR, Murray C, LeClair M, Gallucci S, King MA et al. |
PubMed |
The Journal of Continuing Education in Nursing. |
2017 |
Estados Unidos |
Inglês |
|
A7 |
Administração de medicamento endovenoso em pacientes de hospital de doenças infectocontagiosas: atuação da enfermagem(20) |
Rocha DMA, Brito EAWS, Souza TLV, Mota RO, Miranda MC, Lima FET. |
CINAHL |
Enfermagem em Foco |
2020 |
Brasil |
Português |
|
A8 |
The I-DECIDED clinical decision-making tool for the assessment and safe removal of a peripheral intravenous catheter: a clinimetric assessment(21) |
Ray-Barruel G, Cooke M, Chopra V, Mitchell M, Rickard CM |
PubMed |
BMJ Open |
2020 |
Austrália |
Inglês |
|
A9 |
Outcomes of a simplified ultrasound-guided intravenous training course for emergency nurses(22) |
Feinsmith S, Huebinger R, Pitts M, Baran E, Haas S. |
CINAHL |
Journal of Emergency Nursing |
2018 |
Estados Unidos |
Inglês |
|
A10 |
Uso de tecnologias na terapia intravenosa: contribuições para uma prática mais segura(23) |
Moreira APA, Escudeiro CL, Christovam BP, Silvino ZR, Carvalho MF, Silva RCL |
CINAHL |
Revista Brasileira de Enfermagem |
2017 |
Brasil |
Português |
|
A11 |
Changes in nurses' knowledge and clinical practice in managing local IV complications following an education intervention(24) |
Bayoumi MMM, Khonji LMA, Gabr WFM |
CINAHL |
British Journal of Nursing. |
2022 |
Estados Unidos |
Inglês |
|
A12 |
Safety verification of a new peripheral intravenous catheter placed in the upper arm vein for administration of drugs with high irritant potential(25) |
Murayama R et al. |
PubMed |
Drug Discoveries & Therapeutics |
2022 |
Japão |
Inglês |
|
A13 |
Design and Implementation of An Intravenous Medication Dispensing Robot(26) |
He Y et al.
|
PubMed |
Institute of Electrical and Electronics Engineers IEEE |
2019 |
China |
Inglês |
|
A14 |
The safety of clinically indicated replacement or routine replacement of peripheral intravenous catheters: A randomized controlled study(27) |
Lu H et al. |
PubMed |
The Journal of Vascular Acces
|
2021 |
Estados Unidos |
Inglês |
|
A15 |
Prática nacional dos profissionais de Enfermagem na inserção de dispositivos de acesso vascular periférico(28) |
Silva BSM, Santos LM, Rocha PK, Mota ANB, Avelar AFM, Kusahara DM |
LILACS |
Revista Latino – Americana de Enfermagem |
2024 |
Brasil |
Português |
|
A16 |
Application of Intelligent Intravenous Drug Dispensing Robot in Clinical Nursing(29) |
Zhang L, Liu W, Zhang Y |
PubMed |
IEEE Transactions on Industrial Electronics |
2022 |
China |
Inglês |
|
A17 |
Making It Stick: Developing and Testing the Difficult Intravenous Access (DIVA) Tool(30) |
PubMed |
AJN, American Journal of Nursing |
2018 |
Estados Unidos |
Inglês |
|
|
A18 |
Implementation and evaluation of short peripheral intravenous catheter flushing guidelines: a stepped wedge cluster randomised trial(31) |
Keogh S et al.
|
PubMed |
BMC Medicine |
2020 |
Austrália |
Inglês |
|
A19 |
Rotulagem de dispositivos de terapia intravenosa em Unidades de Terapia Intensiva: revisão integrativa(32) |
Nunes GK, Campos JF, Silva RC. |
LILACS |
Revista Brasileira de Enfermagem |
2022 |
Brasil |
Português |
|
A20 |
The Efficacy, Safety, and Convenience of a New Device for Flushing Intravenous Catheters (Baro Flush™): A Prospective Study(33) |
Choi YI et al. |
PubMed |
Medicina (Kaunas) |
2020 |
Coreia |
Inglês |
|
A21 |
Changes in medication administration error rates associated with the introduction of electronic medication systems in hospitals: a multisite controlled before and after study(34) |
Westbrook JI et al. |
PubMed |
BMJ Health & Care Informatics |
2020 |
Austrália |
Inglês |
|
A22 |
Canulação do acesso vascular em pacientes em hemodiálise: abordagem técnica(35) |
Castro MCM et al. |
LILACS |
Brazilian Journal of Nephrology |
2020 |
Brasil |
Português |
|
A23 |
Comparison of Standard Technique, Ultrasonography, and Near-Infrared Light in Difficult Peripheral Vascular Access: A Randomized Controlled Trial(36) |
Yalçinli SMD, Akarca FK, Can MDO, Uz MDI, Konakçi G
|
Web of Science |
Prehospital and Disaster Medicine |
2022 |
Estados Unidos |
Inglês |
|
Radial artery cannulation in intensive care unit patients: Does distance from wrist joint increase catheter durability and functionality? (37) |
Imbriaco G, Monesi A, Giugni A, Cilloni N |
Web of Science |
The Journal of Vascular Access |
2021 |
Itália |
Inglês |
Source: Research data (2024).
|
Article number |
Objective |
Method |
Sample type |
Type of data analysis |
|
A1 |
To demonstrate the applicability of the Healthcare Failure Mode and Effect Analysis tool for proactively analyzing the risks related to the insertion, maintenance, and removal of peripheral intravenous catheters.. |
Theoretical experimental study |
Intentional non-probabilistic |
Quantitative |
|
A2 |
Based on an evaluation of the nursing team's flushing practices, develop a prototype care technology aimed at promoting best practices in the maintenance of intravenous catheters in intensive care. |
Methodological study of technology development. |
Non-probabilistic, for convenience. |
Descriptive quantitative |
|
A3 |
Identify the dynamics of medication administration. Develop a Standard Operating Procedure for intravenous medication administration. Develop a Checklist with the steps to be followed for safe medication administration to assess adherence by professionals. |
Descriptive study |
Intentional non-probabilistic |
Qualitative |
|
A4 |
To evaluate ultrasound-guided peripheral intravenous (IV) catheterization, compare it with conventional catheterization, and analyze patient pain and satisfaction with catheterization. |
Randomized controlled clinical trial |
Intentional non-probabilistic |
Descriptive quantitative |
|
A5 |
Validate the content of the Standard Operating Procedure for nursing intervention with patients using vasoactive drugs in the emergency room. |
Methodological validation study |
Intentional non-probabilistic |
Quantitative |
|
A6 |
To describe a collaboration between private industry and a hospital to modify, implement, and evaluate a simulation-based combined PIVC insertion continuing education program for nursing staff. |
Randomized crossover clinical trial |
Probabilistic |
Quantitative-qualitative |
|
A7 |
To evaluate the practice of administering intravenous medications to adult patients hospitalized in a hospital for infectious diseases. |
Cross-sectional observational study |
Intentional non-probabilistic |
Descriptive quantitative |
|
A8 |
To describe the clinimetric validation of the I-DECIDED tool for assessment and decision-making regarding peripheral intravenous catheters. |
Methodological validation study |
Intentional non-probabilistic |
Quantitative-qualitative
|
|
A9 |
To implement a simplified, low-cost ultrasound-guided intravenous access (USGIV) training program for emergency nurses that would require a 4-hour time commitment and examine the impact of this program on intravenous attempts in patients. |
Single-arm prospective clinical trial |
Non-probabilistic for convenience |
Quantitative |
|
A10 |
To identify the difficulties faced by the nursing team in managing technologies during intravenous therapy (IV) and to discuss the identified difficulties from the perspective of patient safety. |
Descriptive study |
Non-probabilistic for convenience |
Qualitative |
|
A11 |
To transfer nurses' theoretical knowledge about the prevention of common local complications of intravenous therapy to clinical practice, improving the care and maintenance of peripheral venous catheters (PVCs). |
Quasi-experimental study |
Non-probabilistic for convenience |
Quantitative |
|
A12 |
To evaluate the safety of the new PIVC in patients undergoing administration of highly irritating non-anticancer medications, assessing the occurrence of adverse events. |
Mixed exploratory study |
Probabilistic |
Quantitative |
|
A13 |
To design, build, and implement a robot for dispensing intravenous medications. |
Applied research study |
Probabilistic |
Quantitative |
|
A14 |
To explore the safety of replacing a peripheral intravenous catheter (PIC) every 96 hours compared to clinically indicated catheter removal. |
Prospective, randomized, controlled clinical trial. |
Non-probabilistic for convenience |
Quantitative |
|
A15 |
To identify and compare the practices of nursing professionals regarding the insertion of peripheral vascular access devices, according to their professional category. |
Cross-sectional study of the descriptive survey type. |
Intentional non-probabilistic |
Qualitative |
|
A16 |
To evaluate the effectiveness and impacts of using a medication dispensing robot compared to manual dispensing, specifically in a hospital setting. |
Observational experimental study |
Intentional non-probabilistic
|
Quantitative |
|
A17 |
To develop, validate, and refine a simple, evidence-based tool that novice nurses can use in the clinical setting to better identify patients with pre-existing conditions or anatomical variations that result in access difficulties. |
Methodological validation study |
Non-probabilistic for convenience
|
Quantitative |
|
A18 |
To evaluate the impact that a multifaceted intervention focused on short-term maintenance of PIVC had on patient outcomes. |
Randomized clinical trial |
Non-probabilistic for convenience |
Quantitative |
|
A19 |
To synthesize the evidence on intravenous device labeling used to identify medications administered to patients in Intensive Care Units, with a view to preventing medication errors. |
Systematic review |
Probabilístic |
Qualitative |
|
A20 |
Evaluate the effectiveness, safety, and convenience of a new flushing technique using a Baro Flush™ controller. |
Estudo clínico prospectivo |
Non-probabilistic for convenience |
Quantitative-qualitative
|
|
A21 |
To evaluate the effectiveness of an educational intervention aimed at healthcare professionals regarding the application of best practices for the prevention of adverse events in hospitalized patients. |
Randomized clinical trial |
Non-probabilistic for convenience |
Qualitative |
|
A22 |
Evaluate the cannulation technique and determine which factors are associated with each detail of the technique. |
Cross-sectional study |
Non-probabilistic for convenience |
Quantitative |
|
A23 |
To compare the standard technique, ultrasound (USG) and near-infrared (NIR) light, in terms of first-attempt success in patients with DVA. |
Prospective cohort study |
Non-probabilistic for convenience
|
Quantitative |
|
A24 |
To analyze some variations commonly applied during different individual approaches by intensivists in performing radial artery cannulation in ICU patients and their possible correlation with a longer period of effective radial artery catheter functionality. |
Prospective cohort study |
Non-probabilistic for convenience |
Quantitative |
Source: Research data (2024).
Regarding the type of study, clinical trial (30%) and methodological (17%) studies predominated. As for the type of sample adopted, the vast majority of studies (50%) used non-probabilistic convenience sampling, followed by non-probabilistic purposive sampling (34%) and, finally, probabilistic sampling (17%). Finally, regarding the data analysis technique, quantitative analysis predominated (67%), followed by qualitative (21%) and mixed-methods (12%).
DISCUSSION
Through the analysis of the selected articles (described in Table 1 and Table 2), three thematic categories were formulated: Category 1 – Evidence-based practices and techniques for the safe administration of intravenous medications; Category 2 – Main errors in intravenous medication administration and prevention and care measures; Category 3 – Educational technologies for teaching and training nursing professionals on intravenous medication administration.
Category 1 – Evidence-based practices and techniques for the safe administration of intravenous medications
Intravenous (IV) medication administration is an essential practice in healthcare. However, this procedure presents significant risks that can compromise patient safety. Combined with this, numerous innovations related to the intravenous administration process are emerging, making it essential for professionals to stay updated on evidence-based practices and techniques to minimize risks and ensure the quality of care provided. In this sense, aiming to present innovations related to the intravenous administration process, articles A4, A10, A12, A13, A16, A17, A18, A20, and A22 discuss new techniques and devices focused on the safe administration of medications, from the moment of checking and distributing the medication to the moment of catheter removal (17, 23, 25, 26, 29, 30, 31, 33, 35).
From this perspective, articles A13, A16, and A19 address the medication labeling and dispensing process, listing the benefits of standardization in reducing errors in the administration process. Article A19 states that, although labeling offers important benefits for safety in medication administration, there is still a significant lack of standardization regarding the content, format, and colors of labels in healthcare institutions. This inconsistency hinders the rapid and accurate identification of medications, compromising safety and increasing the risk of errors during preparation and administration (26,29,32).
Along these lines, articles A13 and A16 propose the introduction of a new technology in the medication dispensing process, which can modify and facilitate this process: medication dispensing robots in the hospital environment, representing a significant advance in terms of efficiency, safety, and precision in the dispensing of intravenous medications. Study A13 indicated that this medication dispensing robot was designed with a modular architecture, in which different modules perform specific functions, such as disinfecting vials, opening ampoules, and mixing liquid or powdered medications in infusion bags. Thus, this robot is equipped with computer vision and an expert system, so that it adjusts its operations according to the specifications of the medical prescription, ensuring high precision in dispensing and reading barcodes. In tests with real prescriptions, the system proved to be highly reliable, completing tasks automatically and minimizing human contact with needles, which reduces the risk of injury and contamination (26).
Study A16 showed that, when comparing the dispensing robot with the manual process, manual dispensing was initially faster due to the robot's setup time. However, after prolonged periods, the robot's efficiency surpassed that of the manual method, as the latter was impacted by staff fatigue. The simultaneous use of two robots further increased productivity (29). These advances highlight the potential of robotics in improving hospital practices, making processes safer, more efficient, and less prone to errors, while reducing staff overload and improving patient outcomes.
Moving on to the medication administration process, studies A4, A12, A17, and A22 discuss the peripheral puncture process, covering everything from catheter selection to the puncture itself, addressing new techniques and pain assessment during this procedure. Study A4 investigated the effectiveness of ultrasound-guided venous puncture compared to the traditional method. Although there were no significant differences in the overall success rate (91.7% versus 89.9%), the guided technique was particularly effective in patients with greater difficulty of access, reducing the number of attempts and increasing patient satisfaction. Complications were minimal and perceived pain was similar in both groups (17).
Furthermore, in study A12, ultrasound-guided Peripheral Venous Catheter (PVC) insertion was reported to have a 100% success rate, with no adverse events during device placement or removal. After the application of rigorous protocols, including antisepsis and daily monitoring, the catheters remained functional for up to seven days (25). In this sense, studies A12 and A4 highlighted the importance of using ultrasound as a tool to increase the accuracy and safety of venous puncture, and it can be adopted in clinical practice to facilitate the medication administration process, with greater efficiency and comfort for the patient.
In turn, study A17 introduced a tool called Difficult Intravenous Access (DIVA), designed to assist novice nurses in identifying patients with greater difficulty in venous access, such as those with non-palpable veins, tattoos, or scars. This tool was structured in two columns ("Yes" and "No"), in which nurses had to record weighted values (0, 1, or 2) for each variable. After marking all variables, the nurse summed the values of the two columns to obtain a total score, with a score of 4 or more indicating a high probability of difficulties in performing the puncture. Thus, the implementation of the tool resulted in increased nurses' confidence and a reduction in insertion attempts, promoting greater efficiency and comfort for patients, thus being an important piece of evidence that can assist in the clinical practice of professionals in performing difficult venous access (30).
Study A22 evaluated the characteristics of vascular access in hemodialysis, the puncture techniques used, and their relationship with the efficiency of dialysis therapy. The study highlights three main methods used for puncture: rope ladder, area, and buttonhole. In this sense, the rope ladder technique is the most recommended, as it uses the entire length of the vessel, reducing complications such as thrombosis and stenosis. This technique requires a large surface area, alternating puncture sites at each session, with a minimum distance of 5 to 7 cm between insertions. The area technique concentrates punctures in a limited region, reducing local pain over time; however, it is associated with a higher risk of complications such as aneurysms and infections. Finally, the buttonhole technique, indicated for patients with short arteriovenous fistulas and home dialysis, is less painful but presents a higher risk of infection. In this sense, the study highlights that the efficiency of dialysis treatment is directly influenced by adjustments in the puncture technique, including the puncture location, needle direction, treatment time, blood flow rate, and bevel positioning (35).
These findings highlight the importance of updating professionals on innovative techniques and devices, such as ultrasound and predictive tools, combined with well-defined training and protocols, to optimize clinical practice. Such strategies contribute to better outcomes, with fewer complications, greater efficiency, and greater patient comfort.
Category 2 – Main errors in intravenous medication administration and prevention and care measures
The practice of intravenous medication administration is a routine, yet complex, process in clinical practice. Therefore, several analyzed studies highlight errors related to the insertion, maintenance, and management of peripheral intravenous devices and peripheral intravenous catheters (PICs), as well as evaluating the effectiveness of interventions and technological systems to improve clinical practice. In this sense, study A1 highlighted potential errors in the insertion and maintenance process of PICs, identifying risks such as inadequate antisepsis, failures in administration technique, and lack of detailed records. To mitigate these problems, measures such as regular training, standardized kits, care audits, and the implementation of evidence-based bundles have been proposed (14).
From the perspective of comparing intravenous catheter insertion techniques, study A23 compared three methods of peripheral intravenous catheter insertion in patients with difficult vascular access (DVA): the standard technique, infrared device (NIR), and ultrasound (USG). The ultrasound method showed a higher success rate on the first attempt (78.9%), while the NIR group (58.9%) required more attempts. The procedure duration was longer in the USG group, but there was no significant difference between the methods regarding the need for subsequent interventions. Thus, ultrasound stood out as a highly effective method compared to the other techniques (36).
Furthermore, study A24, when analyzing the radial artery puncture technique, revealed that ultrasound (USG) plays an essential role in optimizing catheter placement and functionality. Among the procedures observed, 68.9% were performed blindly and 31.1% with ultrasound. Furthermore, it was shown that catheters positioned between 4 and 10 cm from the wrist, mainly with ultrasound, presented twice the in situ time compared to those inserted blindly. Thus, the study showed that puncture in the proximal forearm, at a distance of at least 4 cm from the wrist, can increase the durability and functionality of the catheter for patients (37).
When analyzing the techniques adopted after catheter insertion, study A2 pointed to the prevalence of errors in the catheter flushing procedure, with only 22% of professionals performing the procedure correctly and the recommended push-pause technique being adopted by only 35%. The study highlights factors such as lack of standardization, insufficient training, and absence of monitoring as causes of these deviations. To address these failures, a practical guide was developed to standardize the flushing technique, with evidence-based guidelines. This guide addresses the recommended push-pause technique, which is based on fluid flow dynamics, consisting of administering 10 short 1 ml boluses of solution, interspersed with brief pauses, so that the interruptions create variations in flow, generating more efficient shear in the removal of solid deposits when compared to a continuous flow of 10 ml, which promotes better fluid dispersion, reducing the risk of biofilm formation and maintaining the functionality of the device for longer (15).
Regarding failures related to intravenous medication administration, study A7 highlighted deficiencies in hand hygiene (34.3%), lack of essential records, and limited communication with patients (24.3%), although all professionals read the prescription (20). Furthermore, study A15 indicated that professionals also had difficulties in the appropriate selection of materials and advanced techniques, such as the use of vascular ultrasound. These gaps underscore the urgent need for training, supervision, and standardization of guideline-based practices (28).
In addition, study A21 analyzed the impact of electronic medication systems, recording a 14% reduction in administration errors and a 56% decrease in serious errors in intervention wards. These electronic medication systems, also called computerized provider order entry systems, allow clinicians to electronically record both the prescription and administration of medications. Thus, the study highlighted that nurses used these systems to identify the medications to be administered to patients, by displaying the medications to be administered on the system screen(34).
Regarding complications after intravenous administration, study A14 highlighted that several complications are likely to occur, notably phlebitis and infiltrations, which result from failures in catheter management, including hygiene, monitoring, and replacement within the indicated time. The study emphasized that comparisons between PIVC replacement methods showed that clinically indicated replacement resulted in a longer dwell time (83.62 hours versus 69.75 hours in the routine replacement method), indicating the need for greater attention in the management of the devices. Furthermore, the study concluded that periodic catheter replacement every 96 hours is safer(27).
In summary, the studies highlight several errors related to intravenous administration, making clear the importance of evidence-based interventions, continuous training, and advanced technologies to improve the safety and effectiveness of intravenous device management. These improvements are fundamental to reducing complications, optimizing resources, and raising the quality of care provided.
Category 3 - Educational technologies for teaching and training nursing professionals on intravenous medication administration
Intravenous medication administration is a common activity in the daily work of nursing professionals, being a complex process that urgently requires immense attention and training of the professionals involved. Given this perspective, the need to use tools to promote the continuous training of these professionals is evident. As argued in articles A1, A3, A6, A10 and A11, the development of educational technologies and training aimed at nursing professionals, especially related to the insertion, maintenance and removal of venous catheters used for medication infusion, leads to a reduction in common risks and complications, as well as promoting greater safety and quality of care directed to the patient (14,16,19,23,24).
In this sense, it is understood that the scope of educational technologies is vast, so that, in relation to the process of administering intravenous medications, articles A6, A9 and A11 highlight tools aimed at improving the technical and theoretical skills of nursing professionals, involving theoretical-practical didactic courses, which include everything from the use of audiovisual technologies in virtual and face-to-face environments, to the use of realistic simulators associated with the process of inserting and maintaining peripheral catheters through which intravenous administration is performed (19,22,24). Furthermore, studies A1, A2, A3, A5 and A8 highlight educational tools that standardize the performance of nursing professionals, with emphasis on Checklists, Standard Operating Procedures (SOPs) and Care Guides (14-16,21).
Focusing on educational technologies aimed at professional development, article A6 emphasizes the application of an educational tool that links the completion of a course in digital modules and the use of practical simulators focused on the process of inserting peripheral intravenous catheters. During the online course, professionals accessed content related to anatomy, catheter selection, insertion techniques, and management of complications of peripheral venous access, while in practical workshops, with haptic simulators and realistic arms, repetitive practice of technical skills was promoted. In summary, it was highlighted that this tool promoted improvements in the theoretical knowledge (31% improvement) and skills (24% improvement) of the professionals, who evaluated the tool as effective and applicable in practice, to promote greater qualification in performing this procedure, with the necessary technical support (19).
Furthermore, studies A9 and A11 present tools similar to the one analyzed previously, involving theoretical-practical training. In this context, article A9 highlighted the promotion of combined training, involving a short theoretical course and practical simulations on training models to empower nurses in ultrasound-guided intravenous puncture (USGIV), targeting patients with difficult venous access. After the training, nurses demonstrated a notable improvement in the success rate of intravenous punctures, increasing from 81% to 96% after practicing 30 punctures, in addition to a reduction in the total number of intravenous access attempts, reflecting greater clinical efficiency (22).
In turn, study A11 presented an educational strategy that involved conducting a theoretical class addressing methods for preventing complications and maintaining catheters, as well as practical demonstrations on mannequins. This approach resulted in significant improvements in clinical practices, such as the appropriate use of transparent dressings (from 15.6% to 60.9%) and the maintenance of aseptic technique (from 78.1% to 100%), as well as advances in patient education, expanding guidance on cannula care and recognition of signs of infection (24).
Regarding educational tools aimed at standardizing nursing professionals' practices in the intravenous administration process, articles A1 and A8 highlighted the application of educational tools adapted to intravenous administration practice. Article A1 addressed the Healthcare Failure Mode and Effect Analysis (HFMEA) tool, related to the analysis of risks and failures in care processes, which was adapted to allow the identification of critical points in the procedures for insertion, maintenance, and removal of intravenous catheters, enabling the creation of action plans to minimize risks. Thus, the application of HFMEA proved to be relevant, as it standardized the assessment of risks associated with hand hygiene, antisepsis of the area to be punctured, disinfection of the connector and care in choosing the puncture site, monitoring and careful removal of the device, facilitating learning and improving procedures regarding the insertion, maintenance and removal of intravenous catheters, promoting patient safety (14).
In this line of thought, article A8 highlighted the implementation of a clinical decision-making tool (I-DECIDED), which was validated by experts and based on scientific evidence, thus guiding and standardizing nursing professionals' decision-making regarding catheter placement, including the evaluation of clinical parameters associated with catheter presence, device necessity, functionality, complications, infection prevention, and the need for health education for the patient/family, aiming to promote greater safety. In this way, it is clear that the use of the I-DECIDED tool allows for the adoption of evidence-based nursing practice in decision-making regarding intravenous catheter care, promoting the early removal of unnecessary or complicated intravenous accesses, in order to prevent painful problems, infections, and unnecessary costs (21).
In this perspective, article A2 highlighted the development of an educational tool in the form of a care guide, associated with the practice of flushing. In this way, the guide was developed based on scientific evidence, providing a set of guidelines on the preparation, volume, frequency, and technique of performing flushing. Furthermore, the guide identifies common errors made during the flushing procedure, offering solutions to minimize them. In this context, it is clear that the application of this guide can allow professionals to follow the same steps and reduce the risk of complications, such as catheter obstruction or infections, promoting the efficiency of nursing care (15).
In addition, article A3 highlighted the development of a Standard Operating Procedure (SOP) and a Checklist as educational tools to standardize the administration of intravenous medications. In this sense, the SOP provides guidance on each step of the process, from understanding the prescription, hand hygiene, medication preparation, labeling of the prepared medication, instructions for the patient, to administration and monitoring after infusion; The checklist acts as a complementary visual resource that synthesizes the information in the SOP and provides greater practicality for adherence to safe practices, including prescription confirmation and venous access assessment. From this perspective, it became clear that the application of these tools enables better organization and minimizes the risks of complications and errors in intravenous administration (16).
Similarly, article A5 aimed to develop and validate an SOP directed at nursing team interventions in the administration of vasoactive drugs. This SOP included common interventions, such as medication identification/labeling with patient information, dosage, date and time, and the professional's name; administration via infusion pump; assessment of catheter patency and frequent verification of vital signs, as well as specific interventions for five types of vasoactive drugs. This tool was validated by 16 participants (13 nurses, two pharmacists and one biomedical scientist), highlighting that its use may contribute to the reduction of errors, resulting in continuous improvements in the safety and quality of care for patients using vasoactive drugs(18).
FINAL CONSIDERATIONS
This study enabled the identification of innovative technologies and evidence-based strategies to improve intravenous medication administration. Significant advances were revealed in the use of technologies, standardized protocols, and educational tools, all aimed at reducing risks and improving the safety, efficiency, and quality of care.
Recognizing the complex and routine nature of intravenous medication administration, the need for regular training for professionals became apparent. The importance of implementing new educational technologies for the training and updating of professionals involved was highlighted, promoting significant improvements in practical skills and theoretical knowledge. Therefore, this study demonstrates the importance of integrating scientific evidence into daily nursing care, ensuring better patient outcomes and contributing to excellence in healthcare.
REFERENCES
- Uramatsu M, Kimura N, Kojima T, Fujisawa Y, Oto T, Barach P. Frontline nursing staff’s perceptions of intravenous medication administration: the first step toward safer infusion processes - a qualitative study. BMJ Open Quality [Internet]. 2024 Jun 1 [cited 2024 Jul 16];13(2):e002809. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC11216072/
- Romero MP, González RB, Calvo MSR, Fachado AA. A segurança do paciente, qualidade do atendimento e ética dos sistemas de saúde. Rev Bioética [Internet]. 2018 Dec;26(3):333–42. Available from: https://www.scielo.br/j/bioet/a/4hRnkzkJFL8MxdRByNv7LPj/?format=pdf&lang=pt.
- World Health Organization. Medication Without Harm - Global Patient Safety Challenge on Medication Safety. Geneva: World Health Organization; 2017.
- Conselho Federal de Enfermagem. Resolução COFEN nº 564/2017. Aprova novo Código de Ética dos Profissionais de Enfermagem. Disponível em: http://www.cofen.gov.br/resolucao-cofen-no-5642017_59145.html.
- Reis UOP, Santos LG, Carvalho GA, et al. Erros no preparo e na administração de medicamentos intravenosos. Rev Baiana Enferm. 2020;34(2):1-11. Available from: https://periodicos.ufba.br/index.php/enfermagem/article/view/36450/34948
- Vilela RPB, Jericó M de C. Implantação de tecnologias para prevenção de erros de medicação em hospital de alta complexidade: análise de custos e resultados. Einstein (São Paulo) [Internet]. 2019 Jul 1;17:eGS4621. Available from: https://www.scielo.br/j/eins/a/56m5gPm38QwGPcNcJYkDcvD/?lang=pt#.
- Ferreira MA, Leandro GB, Fernandes MC, Rolim ALG, Andrade ME de. Tecnologias educacionais no empoderamento do adolescente acerca da depressão. Revista de Enfermagem UFPE on line [Internet]. 2019 Jan 3 [cited 2022 Jul 4];13(1):275–80. Available from: https://periodicos.ufpe.br/revistas/revistaenfermagem/article/view/237881/31175.
- Galvão RO, Teixeira E, Nemer CRB. Guia ilustrado para mediar educação em saúde com pessoas após o acidente vascular cerebral: construção e validação de conteúdo. Rev Eletrôn Acervo Saúde. 2020;12(11):1-11. Available from: https://acervomais.com.br/index.php/saude/article/view/4450/2575.
- Sousa LB de, Braga HFGM, Alencastro ASA, Silva MJN da, Oliveira BSB de, Santos LVF dos, et al. Effect of educational video on newborn care for the knowledge of pregnant and postpartum women and their families. Rev Bras Enfermagem. 2022;75(suppl 2). Available from: https://www.scielo.br/j/reben/a/Hpvqw8JGwbNt5jxMjdTYz6M/?lang=en
- Souza-Junior VDD, Mendes IAC, Tori R, Marques LP, Mashuda FKK, Hirano LAF, et al. VIDA-Nursing v1.0: immersive virtual reality in vacuum blood collection among adults. Revista Latino-Americana de Enfermagem [Internet]. 2020 Jun 1;28. Available from: https://www.scielo.br/j/rlae/a/nv5Y6MXbWrFCmRZy5XfcfMy/abstract/?lang=en.
- Peters MDJ, Godfrey C, McInerney P, Khalil H, Larsen P, Marnie C, et al. Best practice guidance and reporting items for the development of scoping review protocols. JBI Evidence Synthesis [Internet]. 2022 Jan 31;20(4):953–68. Available from: https://journals.lww.com/jbisrir/fulltext/2022/04000/Best_practice_guidance_and_reporting_items_for_the.3.aspx.
- Mattos SM, Cestari VRF, Moreira TMM. Scoping protocol review: PRISMA-ScR guide refinement. Rev Enferm UFPI. 2023;12(1):1–12. Disponível em: https://periodicos.ufpi.br/index.php/reufpi/article/view/3062/3689.
- Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A. Rayyan - a web and mobile app for systematic reviews. Syst Rev. 2016;5(1):210. Disponível em: https://systematicreviewsjournal.biomedcentral.com/articles/10.1186/s13643-016-0384-4.
- Furlan MS, Marques RM, Cavalcante RB, Oliveira PP, Castro M. Process of insertion, maintenance and removal of peripheral intravenous catheters: preventive risk analysis. Cogitare Enferm. 2023;28(1):1–13. Disponível em: https://dx.doi.org/10.1590/ce.v28i0.91553.
- Ribeiro GSR, Marques IA, Santos TM, Oliveira RS, Santos TM, Gouveia BR. Protótipo de tecnologia de cuidado para prática do flushing na manutenção de cateteres intravenosos. Texto Contexto Enferm. 2023;32(1):1–16. Disponível em: https://doi.org/10.1590/1980-265X-TCE-2022-0302pt.
- Alves SZSP. Administração segura de medicamentos via endovenosa: uma proposta para o hospital universitário [dissertação]. Universidade Federal do Estado do Rio de Janeiro; Rio de Janeiro (RJ) 2017. Disponível em: https://docs.bvsalud.org/biblioref/2019/12/1026808/sarah-zani-sampaio-piraciaba-alves_relatorio-de-pesquisa.pdf.
- Salleras-Duran L, Munoz E, Garcia R, Casajuana J, Uson J, Sanchez M, et al. Pain and Satisfaction Perceptions of Ultrasound-Guided Versus Conventional Peripheral Intravenous Catheterization: A Randomized Controlled Trial. Pain Manag Nurs. 2024;25(1):37–44. Disponível em: https://www.sciencedirect.com/science/article/pii/S1524904223001546.
- Paim AE, Oliveira BGRB, Bousso RS, Silva LFS. Validation of an instrument regarding nursing intervention in patients in vasoactive therapy. Rev Bras Enferm. 2017;70(3):453–60. Disponível em: http://dx.doi.org/10.1590/0034-7167-2016-0254.
- Glover KR, Brown B, Fairbanks RJ. A Simulation-Based Blended Curriculum for Short Peripheral Intravenous Catheter Insertion: An Industry–Practice Collaboration. J Contin Educ Nurs. 2017;49(9):397–406. Disponível em: https://pubmed.ncbi.nlm.nih.gov/28850656/.
- Rocha DMA, Oliveira VB, Araújo STC, Silva AN, Nascimento DS, Guimarães EMP. Administração de medicamentos endovenoso em pacientes de hospital de doenças infectocontagiosas: atuação da enfermagem. Enferm Foco. 2020;11(2). Disponível em: http://revista.cofen.gov.br/index.php/enfermagem/article/view/2643.
.
- Ray-Barruel G, Cooke M, Slater K, Rickard CM. The I-DECIDED clinical decision-making tool for peripheral intravenous catheter assessment and safe removal: a clinimetric evaluation. BMJ Open. 2020;10(1):e035239. Disponível em: https://bmjopen.bmj.com/content/10/1/e035239.
- Feinsmith S, Huebinger R, Pitts M, Baran E, Haas S. Outcomes of a Simplified Ultrasound-Guided Intravenous Training Course for Emergency Nurses. J Emergency Nursing. 2018 Mar;44(2):169-175.e2. Disponível em: https://dx.doi.org/10.1016/j.jen.2017.10.001.
- Moreira APA, Escudeiro CL, Christovam BP, Silvino ZR, Carvalho MF de, Silva RCL da. Use of technologies in intravenous therapy: contributions to a safer practice. Rev Bras Enfermagem. 2017 Jun;70(3):595–601. Disponível em: https://www.scielo.br/j/reben/a/zvL377WqNqmjCPcZZpXdbzs/?lang=pt.
- Bayoumi MMM, Khonji LMA, Gabr WFM. Changes in nurses’ knowledge and clinical practice in managing local IV complications following an education intervention. Br J Nurs. 2022;31(8):24–30. Disponível em: https://doi.org/10.12968/bjon.2022.31.8.S24.
- Murayama R, Oyama H, Abe-Doi M, Yosuke Masamoto, Kashiwabara K, Tobe H, et al. Safety verification of a new peripheral intravenous catheter placed in the upper arm vein for administration of drugs with high irritant potential. Drug Discoveries Therapeutics [Internet]. 2022 Jun 24;16(3):128–34. Available from: https://pubmed.ncbi.nlm.nih.gov/35753769/
- He Y, Hu Y, Fu X, Cheng X, Jin H, Zhang P. Design and Implementation of An Intravenous Medication Dispensing Robot. CBS. 2019 Sep 1; Disponível em:doi:10.1109/cbs46900.2019.9114514.
- Lu H, Yang Q, Nor HM, Lv Y, Zheng X, Xin X, et al. The safety of clinically indicated replacement or routine replacement of peripheral intravenous catheters: A randomized controlled study. J Vascular Access. 2021 Mar 12;23(3):112972982199852. Disponível em: doi:10.1177/1129729821998528.
- Silva BSM, Santos LM dos, Rocha PK, Mota ANB, Avelar AFM, Kusahara DM. National practice of Nursing professionals in the insertion of peripheral vascular access devices. Rev Latino-Americana de Enfermagem [Internet]. 2024;32. Available from: https://www.scielo.br/j/rlae/a/F5wCLM6wgc9KJqnXqhbtjYd/?lang=pt.
- Zhang L, Liu W, Zhang Y. Application of intelligent intravenous drug dispensing robot in clinical nursing. Contrast Media Mol Imaging. 2022;2022:e4769883. Disponível em: https://onlinelibrary.wiley.com/doi/10.1155/2022/4769883.
- Smith B, Givens K. Making it stick: Developing and testing the difficult intravenous access (DIVA) tool. Am J Nurs. 2018;118(7):56–62. Disponível em: doi:10.1097/01.NAJ.0000541440.91369.00.
- Keogh S, Shelverton C, Flynn J, Mihala G, Mathew S, Davies KM, et al. Implementation and evaluation of short peripheral intravenous catheter flushing guidelines: a stepped wedge cluster randomised trial. BMC Medicine. 2020 Sep 30;18(1). Disponível em: https://bmcmedicine.biomedcentral.com/articles/10.1186/s12916-020-01728-1.
- Nunes G, Campos JF, Celestino R. Rotulagem de dispositivos de terapia intravenosa em Unidades de Terapia Intensiva: revisão integrativa. Rev Bras Enferm. 2022;75(6). Disponível em: https://doi.org/10.1590/0034-7167-2022-0049pt.
- Choi YI, Cho JH, Chung JW, Kim KO, Kwon KA, Chun HY, et al. The Efficacy, Safety, and Convenience of a New Device for Flushing Intravenous Catheters (Baro FlushTM): A Prospective Study. Medicina. 2020 Aug 5;56(8):393. Disponível em: https://www.mdpi.com/1648-9144/56/8/393
- Westbrook JI, Sunderland NS, Woods A, Raban MZ, Gates P, Li L. Changes in medication administration error rates associated with the introduction of electronic medication systems in hospitals: a multisite controlled before and after study. BMJ Health Care Informatics. 2020 Aug;27(3):e100170. Disponível em: https://pmc.ncbi.nlm.nih.gov/articles/PMC7430327/.
- Castro MCM, Carlquist FTY, Silva C de F, Xagoraris M, Centeno JR, Souza JAC de. Vascular access cannulation in hemodialysis patients: technical approach. Braz J Nephrology. 2020 Mar;42(1):38–46. Disponível em: https://doi.org/10.1590/2175-8239-JBN-2019-0031
- Yalçınlı S, Karbek Akarca F, Can Ö, Uz İ, Konakçı G. Comparison of Standard Technique, Ultrasonography, and Near-Infrared Light in Difficult Peripheral Vascular Access: A Randomized Controlled Trial. Prehospital Disaster Medicine. 2021 Dec 6;37(1):65–70. Disponível em: https://www.cambridge.org/core/journals/prehospital-and-disaster-medicine/article/comparison-of-standard-technique-ultrasonography-and-nearinfrared-light-in-difficult-peripheral-vascular-access-a-randomized-controlled-trial/0F3549441C7A58CC4AFC69E2217D83A3.
- Imbriaco G, Monesi A, Giugni A, Cilloni N. Radial artery cannulation in intensive care unit patients: Does distance from wrist joint increase catheter durability and functionality? J Vascular Access. 2020 Aug 27;22(4):561–7. Disponível em: https://journals.sagepub.com/doi/abs/10.1177/1129729820953020.
Funding and Acknowledgments:
This work was carried out with the support of the Coordination for the Improvement of Higher Education Personnel (CAPES), through the granting of Scientific Initiation scholarships, in the PIBIC modality, within the scope of the Federal University of Campina Grande.
Data availability statement
No databases were generated in this study. The information presented is described in the body of the article.
Authorship Criteria (Authors' Contributions)
José Mateus Ismael Lima: Contributed substantially to the conception and planning of the study; to the obtaining, analysis, and interpretation of the data; as well as to the writing.
Fernanda Eli Dantas Gondim: Contributed substantially to the conception and planning of the study; to the obtaining, analysis, and interpretation of the data; as well as to the writing.
Bernadete de Lourdes André Gouveia: Contributed to the writing and critical review.
Maria Cristina Lins Oliveira Frazão: Contributed to the writing and critical review.
Luana Carla Santana Ribeiro: Contributed substantially to the conception and planning of the study; to the obtaining, analysis, and interpretation of the data; as well as to the writing and critical review.
Declaration of conflict of interest
Nothing to declare.
Scientific Editor: Ítalo Arão Pereira Ribeiro. Orcid: https://orcid.org/0000-0003-0778-1447
Rev Enferm Atual In Derme 2026;100(2): e026051